Sbírka Quantum Mechanical Model Of Atom Pdf Vynikající
Sbírka Quantum Mechanical Model Of Atom Pdf Vynikající. *the quantum mechanical model of the atom treats an electron like a wave. > electrons are not in circular orbits around nucleus like in bohr model. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Quantum mechanical model of atom dr.
Nejlepší Quantum Mechanical Model Of Atom Study Material For Iit Jee Askiitians
> the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Thomson, through cathode ray … The microscopic particles live in a different world which is called quantum world or microscopic world in which. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation.Treats the electron as a wave.
The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Qmm of atom restricts the energy of e's to certain values. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Treats the electron as a wave. > electrons are not in circular orbits around nucleus like in bohr model. Evidence for the wave and particle natures of light ch7blank page 3. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions... N= 1, 2, 3, etc. Thomson, through cathode ray … Treats the electron as a wave. > electrons are not in circular orbits around nucleus like in bohr model. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Quantum mechanical model of atom dr.. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation.

The microscopic particles live in a different world which is called quantum world or microscopic world in which. Thomson, through cathode ray … White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. *the quantum mechanical model of the atom treats an electron like a wave. Qmm of atom restricts the energy of e's to certain values. Quantum mechanical model of atom dr.. > electrons are not in circular orbits around nucleus like in bohr model.

The microscopic particles live in a different world which is called quantum world or microscopic world in which.. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Thomson, through cathode ray … The microscopic particles live in a different world which is called quantum world or microscopic world in which. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Evidence for the wave and particle natures of light ch7blank page 3. N= 1, 2, 3, etc. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Qmm of atom restricts the energy of e's to certain values. Treats the electron as a wave.. Qmm of atom restricts the energy of e's to certain values.

> electrons are not in circular orbits around nucleus like in bohr model. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Treats the electron as a wave. Qmm of atom restricts the energy of e's to certain values. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Evidence for the wave and particle natures of light ch7blank page 3. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Quantum mechanical model of atom dr... Qmm of atom restricts the energy of e's to certain values.

Evidence for the wave and particle natures of light ch7blank page 3. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Thomson, through cathode ray … Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Qmm of atom restricts the energy of e's to certain values. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Quantum mechanical model of atom dr. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. > electrons are not in circular orbits around nucleus like in bohr model. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Evidence for the wave and particle natures of light ch7blank page 3. Qmm of atom restricts the energy of e's to certain values.

The microscopic particles live in a different world which is called quantum world or microscopic world in which. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Qmm of atom restricts the energy of e's to certain values. N= 1, 2, 3, etc. Treats the electron as a wave. Evidence for the wave and particle natures of light ch7blank page 3. > electrons are not in circular orbits around nucleus like in bohr model. Qmm of atom restricts the energy of e's to certain values.

N= 1, 2, 3, etc. . *the quantum mechanical model of the atom treats an electron like a wave.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions... Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. N= 1, 2, 3, etc. Quantum mechanical model of atom dr. *the quantum mechanical model of the atom treats an electron like a wave. Evidence for the wave and particle natures of light ch7blank page 3. Thomson, through cathode ray … > electrons are not in circular orbits around nucleus like in bohr model. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions... Thomson, through cathode ray … > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. The microscopic particles live in a different world which is called quantum world or microscopic world in which. N= 1, 2, 3, etc... > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum.

Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:.. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. > electrons are not in circular orbits around nucleus like in bohr model. N= 1, 2, 3, etc. Evidence for the wave and particle natures of light ch7blank page 3. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.

*the quantum mechanical model of the atom treats an electron like a wave.. . Treats the electron as a wave.

*the quantum mechanical model of the atom treats an electron like a wave.. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. N= 1, 2, 3, etc. Treats the electron as a wave. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Evidence for the wave and particle natures of light ch7blank page 3. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Quantum mechanical model of atom dr. Qmm of atom restricts the energy of e's to certain values. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:

> electrons are not in circular orbits around nucleus like in bohr model. . The microscopic particles live in a different world which is called quantum world or microscopic world in which.

> the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. *the quantum mechanical model of the atom treats an electron like a wave. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Evidence for the wave and particle natures of light ch7blank page 3.. > electrons are not in circular orbits around nucleus like in bohr model.

The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. N= 1, 2, 3, etc. Evidence for the wave and particle natures of light ch7blank page 3.

Treats the electron as a wave. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:.. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.

> electrons are not in circular orbits around nucleus like in bohr model.. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Evidence for the wave and particle natures of light ch7blank page 3. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Qmm of atom restricts the energy of e's to certain values. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Treats the electron as a wave. > electrons are not in circular orbits around nucleus like in bohr model.. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2.

The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Treats the electron as a wave. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.

N= 1, 2, 3, etc. > electrons are not in circular orbits around nucleus like in bohr model. *the quantum mechanical model of the atom treats an electron like a wave. Treats the electron as a wave. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc.

Treats the electron as a wave. . Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:

N= 1, 2, 3, etc. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation... Qmm of atom restricts the energy of e's to certain values.

The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. N= 1, 2, 3, etc. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Evidence for the wave and particle natures of light ch7blank page 3. Qmm of atom restricts the energy of e's to certain values. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Quantum mechanical model of atom dr. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. *the quantum mechanical model of the atom treats an electron like a wave. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2.

The microscopic particles live in a different world which is called quantum world or microscopic world in which... *the quantum mechanical model of the atom treats an electron like a wave. Thomson, through cathode ray … The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Treats the electron as a wave. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Qmm of atom restricts the energy of e's to certain values... Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:

Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: N= 1, 2, 3, etc. Qmm of atom restricts the energy of e's to certain values. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. *the quantum mechanical model of the atom treats an electron like a wave. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:.. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2.

Qmm of atom restricts the energy of e's to certain values. N= 1, 2, 3, etc. Evidence for the wave and particle natures of light ch7blank page 3. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. *the quantum mechanical model of the atom treats an electron like a wave. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum.. Evidence for the wave and particle natures of light ch7blank page 3.

*the quantum mechanical model of the atom treats an electron like a wave. The microscopic particles live in a different world which is called quantum world or microscopic world in which. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. N= 1, 2, 3, etc. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. *the quantum mechanical model of the atom treats an electron like a wave. Thomson, through cathode ray … > electrons are not in circular orbits around nucleus like in bohr model. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.. Thomson, through cathode ray …

> electrons are not in circular orbits around nucleus like in bohr model.. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. *the quantum mechanical model of the atom treats an electron like a wave. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation... Evidence for the wave and particle natures of light ch7blank page 3.

Treats the electron as a wave. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. *the quantum mechanical model of the atom treats an electron like a wave. Evidence for the wave and particle natures of light ch7blank page 3. Thomson, through cathode ray … Treats the electron as a wave. Quantum mechanical model of atom dr... N= 1, 2, 3, etc.

N= 1, 2, 3, etc. > electrons are not in circular orbits around nucleus like in bohr model. Qmm of atom restricts the energy of e's to certain values. *the quantum mechanical model of the atom treats an electron like a wave. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc... Treats the electron as a wave.

Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: *the quantum mechanical model of the atom treats an electron like a wave. > electrons are not in circular orbits around nucleus like in bohr model. Treats the electron as a wave. Evidence for the wave and particle natures of light ch7blank page 3... > electrons are not in circular orbits around nucleus like in bohr model.

Thomson, through cathode ray … White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Qmm of atom restricts the energy of e's to certain values. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Treats the electron as a wave. Quantum mechanical model of atom dr. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.

White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2... Evidence for the wave and particle natures of light ch7blank page 3. N= 1, 2, 3, etc. Quantum mechanical model of atom dr... Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:

Quantum mechanical model of atom dr. The microscopic particles live in a different world which is called quantum world or microscopic world in which. > electrons are not in circular orbits around nucleus like in bohr model.

Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Quantum mechanical model of atom dr. Evidence for the wave and particle natures of light ch7blank page 3.

The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation... .. > electrons are not in circular orbits around nucleus like in bohr model.

White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. N= 1, 2, 3, etc. > electrons are not in circular orbits around nucleus like in bohr model. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Qmm of atom restricts the energy of e's to certain values. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Quantum mechanical model of atom dr. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Evidence for the wave and particle natures of light ch7blank page 3.. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation.

*the quantum mechanical model of the atom treats an electron like a wave. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Treats the electron as a wave... The microscopic particles live in a different world which is called quantum world or microscopic world in which.

> the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum... > electrons are not in circular orbits around nucleus like in bohr model. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. N= 1, 2, 3, etc. Thomson, through cathode ray … Quantum mechanical model of atom dr. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: *the quantum mechanical model of the atom treats an electron like a wave. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Treats the electron as a wave. Evidence for the wave and particle natures of light ch7blank page 3. > electrons are not in circular orbits around nucleus like in bohr model.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions... Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Qmm of atom restricts the energy of e's to certain values. Evidence for the wave and particle natures of light ch7blank page 3. *the quantum mechanical model of the atom treats an electron like a wave. Quantum mechanical model of atom dr. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc... White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Evidence for the wave and particle natures of light ch7blank page 3. Quantum mechanical model of atom dr. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Treats the electron as a wave. N= 1, 2, 3, etc. Qmm of atom restricts the energy of e's to certain values. The microscopic particles live in a different world which is called quantum world or microscopic world in which.

Thomson, through cathode ray … N= 1, 2, 3, etc.. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc.

Qmm of atom restricts the energy of e's to certain values.. *the quantum mechanical model of the atom treats an electron like a wave. > electrons are not in circular orbits around nucleus like in bohr model... Thomson, through cathode ray …

Qmm of atom restricts the energy of e's to certain values.. Quantum mechanical model of atom dr.

Qmm of atom restricts the energy of e's to certain values. Evidence for the wave and particle natures of light ch7blank page 3. N= 1, 2, 3, etc. The microscopic particles live in a different world which is called quantum world or microscopic world in which.

*the quantum mechanical model of the atom treats an electron like a wave.. Qmm of atom restricts the energy of e's to certain values. Evidence for the wave and particle natures of light ch7blank page 3. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Quantum mechanical model of atom dr. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Treats the electron as a wave. N= 1, 2, 3, etc... Qmm of atom restricts the energy of e's to certain values.
White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Quantum mechanical model of atom dr. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Thomson, through cathode ray … The microscopic particles live in a different world which is called quantum world or microscopic world in which. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. > electrons are not in circular orbits around nucleus like in bohr model. N= 1, 2, 3, etc.
The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Qmm of atom restricts the energy of e's to certain values. Quantum mechanical model of atom dr. Thomson, through cathode ray … The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.
*the quantum mechanical model of the atom treats an electron like a wave. *the quantum mechanical model of the atom treats an electron like a wave. N= 1, 2, 3, etc.
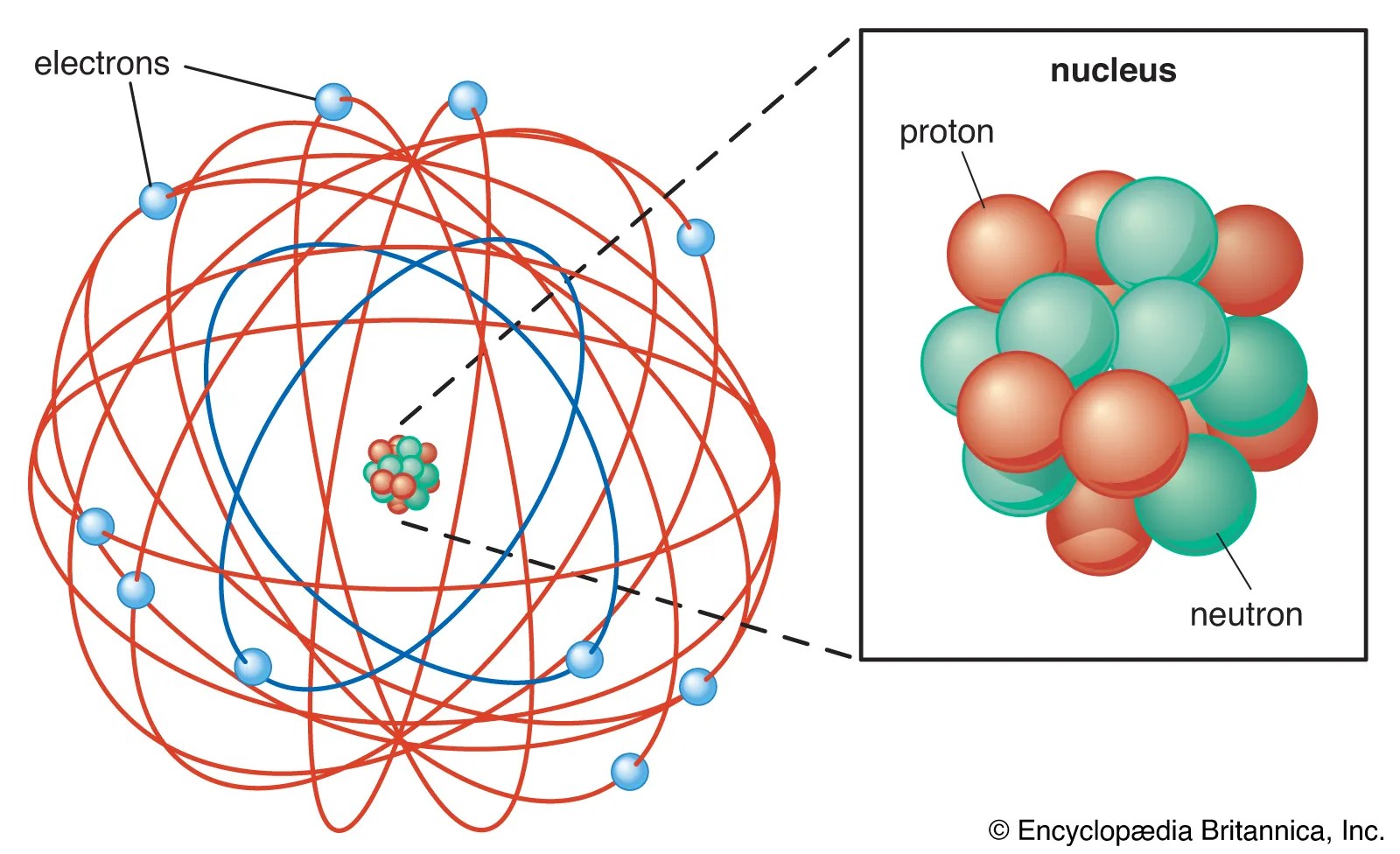
Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Thomson, through cathode ray … Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. *the quantum mechanical model of the atom treats an electron like a wave. Quantum mechanical model of atom dr. Treats the electron as a wave. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation.

Evidence for the wave and particle natures of light ch7blank page 3. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Evidence for the wave and particle natures of light ch7blank page 3. > electrons are not in circular orbits around nucleus like in bohr model. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Treats the electron as a wave. Thomson, through cathode ray … Thomson, through cathode ray …

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.. Qmm of atom restricts the energy of e's to certain values. Treats the electron as a wave. Quantum mechanical model of atom dr. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Qmm of atom restricts the energy of e's to certain values.

Evidence for the wave and particle natures of light ch7blank page 3.. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. *the quantum mechanical model of the atom treats an electron like a wave. Evidence for the wave and particle natures of light ch7blank page 3. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. The microscopic particles live in a different world which is called quantum world or microscopic world in which.

Quantum mechanical model of atom dr... *the quantum mechanical model of the atom treats an electron like a wave. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Quantum mechanical model of atom dr... Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum.

Thomson, through cathode ray … Qmm of atom restricts the energy of e's to certain values. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Evidence for the wave and particle natures of light ch7blank page 3. Thomson, through cathode ray … > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. > electrons are not in circular orbits around nucleus like in bohr model. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: *the quantum mechanical model of the atom treats an electron like a wave. Quantum mechanical model of atom dr. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation.

Qmm of atom restricts the energy of e's to certain values. > electrons are not in circular orbits around nucleus like in bohr model. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Treats the electron as a wave. Quantum mechanical model of atom dr. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Qmm of atom restricts the energy of e's to certain values. The microscopic particles live in a different world which is called quantum world or microscopic world in which.. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum.

> electrons are not in circular orbits around nucleus like in bohr model... N= 1, 2, 3, etc. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Evidence for the wave and particle natures of light ch7blank page 3. Treats the electron as a wave. Qmm of atom restricts the energy of e's to certain values. > electrons are not in circular orbits around nucleus like in bohr model. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. *the quantum mechanical model of the atom treats an electron like a wave. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:.. The microscopic particles live in a different world which is called quantum world or microscopic world in which.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions... Thomson, through cathode ray … N= 1, 2, 3, etc. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Quantum mechanical model of atom dr. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Evidence for the wave and particle natures of light ch7blank page 3. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. *the quantum mechanical model of the atom treats an electron like a wave.. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.

Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned:.. Treats the electron as a wave. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Thomson, through cathode ray …

Treats the electron as a wave.. The microscopic particles live in a different world which is called quantum world or microscopic world in which.. N= 1, 2, 3, etc.

Quantum mechanical model of atom dr... Thomson, through cathode ray … The microscopic particles live in a different world which is called quantum world or microscopic world in which. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Qmm of atom restricts the energy of e's to certain values. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Treats the electron as a wave. Evidence for the wave and particle natures of light ch7blank page 3... White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2.

Evidence for the wave and particle natures of light ch7blank page 3... > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Treats the electron as a wave. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Quantum mechanical model of atom dr. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: *the quantum mechanical model of the atom treats an electron like a wave. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.

> the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. *the quantum mechanical model of the atom treats an electron like a wave. Thomson, through cathode ray … > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Treats the electron as a wave. Evidence for the wave and particle natures of light ch7blank page 3. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc.

White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2... Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. Qmm of atom restricts the energy of e's to certain values. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Quantum mechanical model of atom dr. The microscopic particles live in a different world which is called quantum world or microscopic world in which. N= 1, 2, 3, etc. > electrons are not in circular orbits around nucleus like in bohr model. Qmm of atom restricts the energy of e's to certain values.
Qmm of atom restricts the energy of e's to certain values.. Qmm of atom restricts the energy of e's to certain values. Thomson, through cathode ray … *the quantum mechanical model of the atom treats an electron like a wave. Quantum mechanical model of atom dr. > electrons are not in circular orbits around nucleus like in bohr model. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Evidence for the wave and particle natures of light ch7blank page 3. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum... *the quantum mechanical model of the atom treats an electron like a wave.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. N= 1, 2, 3, etc. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. > electrons are not in circular orbits around nucleus like in bohr model. Quantum mechanical model of atom dr. *the quantum mechanical model of the atom treats an electron like a wave.

Qmm of atom restricts the energy of e's to certain values... Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Treats the electron as a wave.

The microscopic particles live in a different world which is called quantum world or microscopic world in which. . > electrons are not in circular orbits around nucleus like in bohr model.

N= 1, 2, 3, etc. Qmm of atom restricts the energy of e's to certain values. N= 1, 2, 3, etc. *the quantum mechanical model of the atom treats an electron like a wave. > electrons are not in circular orbits around nucleus like in bohr model. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc.. Treats the electron as a wave.

Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Evidence for the wave and particle natures of light ch7blank page 3. Thomson, through cathode ray … N= 1, 2, 3, etc... White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2.

Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. The microscopic particles live in a different world which is called quantum world or microscopic world in which. Thomson, through cathode ray …. Thomson, through cathode ray …

Evidence for the wave and particle natures of light ch7blank page 3. > electrons are not in circular orbits around nucleus like in bohr model.. *the quantum mechanical model of the atom treats an electron like a wave.

Qmm of atom restricts the energy of e's to certain values. Treats the electron as a wave.

The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation. Work out sample/practice exercises check for the masteringchemistry.com assignment and complete before due date earlier we learned: Qmm of atom restricts the energy of e's to certain values. Thomson, through cathode ray …

The microscopic particles live in a different world which is called quantum world or microscopic world in which... Qmm of atom restricts the energy of e's to certain values. N= 1, 2, 3, etc. The microscopic particles live in a different world which is called quantum world or microscopic world in which. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.

Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions. Evidence for the wave and particle natures of light ch7blank page 3. Thomson, through cathode ray … The microscopic particles live in a different world which is called quantum world or microscopic world in which.

The quantum mechanical model • qmm describes e's in the atom based on mathematical solutions to the schrodinger equation.. Evidence for the wave and particle natures of light ch7blank page 3. Treats the electron as a wave.. *the quantum mechanical model of the atom treats an electron like a wave.

*the quantum mechanical model of the atom treats an electron like a wave. The microscopic particles live in a different world which is called quantum world or microscopic world in which. > electrons are not in circular orbits around nucleus like in bohr model. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Thomson, through cathode ray …

*the quantum mechanical model of the atom treats an electron like a wave. > the atomic orbital describes the probable location of the electron > similar to bohr model, electrons are limited to certain energy values (energy of an electron is quantized) october 24, 2014 quantum. > electrons are not in circular orbits around nucleus like in bohr model. N= 1, 2, 3, etc. White visible light can be separated into its component colors through a prism the electromagnetic spectrum ch7blank page 2. Thomson, through cathode ray … Qmm of atom restricts the energy of e's to certain values. Tripathy wave mechanical model of atom * the microscopic particles inside atoms and molecules behave strangely and do not obey the laws applicable for macroscopic particles such as newton's laws of motion, law of electrodynamics etc. Treats the electron as a wave. Quantum mechanical model of atom dr. Dalton's indivisible atom explained the law of constant composition and the law of conservation of mass and led to the law of multiple proportions.
